Shop
GARDpotency
GARD®potency, OECD TGP 4.106
Skin sensitizing potency classification according to GHS/CLP
GARDpotency is an in vitro add-on test to GARDskin for potency classification according to GHS/CLP. The GARDpotency signature comprises 51 genes. The assay can subcategorize sensitizers that have been identified by GARDskin into strong sensitizers (1A) or weak sensitizers (1B).
GARDpotency is included in the OECD Test Guideline Program (TGP 4.106) and currently undergoing ESAC peer-review. Already, GARDpotency results can be used as weight-of-evidence in REACH dossiers for sub-categorization of confirmed skin sensitizers into 1A or 1B according to the GHS/CLP system.
How it works:
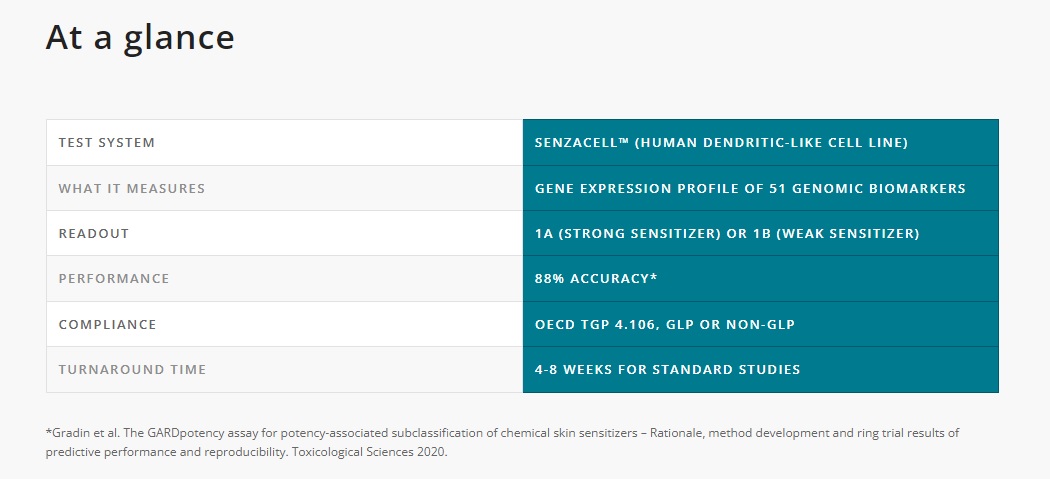
GARDpotency is an add-on test to GARDskin. They are performed in a tiered approach for GHS/CLP potency classification. In the first tier, a test sample is classified by GARDskin as either skin sensitizer or non-sensitizer. If the test sample is classified as a skin sensitizer, it then can be further classified by GARDpotency in the second tier testing. As a result, the test sample can be sub-categorized as 1A (strong) or 1B (weak) skin sensitizer according to the GHS/CLP system.
Contact us for a quotation.